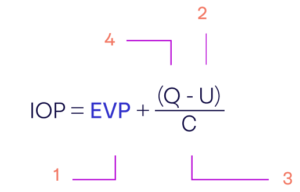
Intraocular pressure is comprised of four distinct components that, when combined, lead to total intraocular pressure (IOP). These four components are currently best described by the modified Goldmann equation, which includes contributions from the Aqueous Humor Inflow Rate (Q), Uveoscleral Outflow Rate (U), Conventional Outflow Facility (C), and Episcleral Venous Pressure (EVP). EVP is the largest component of IOP, contributing around 2/3 of total IOP. However, no approved therapy to date selectively targets EVP.

QLS-111 is the only therapy targeting EVP
Episcleral Venous Pressure
- Selectively targeted by QLS-111
- Contributes approximately 2/3 of total IOP
Uveoscleral Outflow Rate
- Latanoprost
- Bimatoprost
- Travaprost
- Tafluprost
- Latanoprostene bunod
- Brimonidine
- Apraclonidine
Conventional Trabecular Outflow Facility
- Pilocarpine
- Carbachol
- Netarsudil
Aqueous Humor Inflow Rate
- Timolol
- Betaxolol
- Levobunolol
- Metipranolol
- Brimonidine
- Apraclonidine
- Brinzolamide
- Dorzolamide
- Acetazolamide
- Methazolamide
Our Pipeline
| Phase 1 | Phase 2 | Phase 3 | Approval | ||||
|---|---|---|---|---|---|---|---|
| QLS-111POAG/OHT | Phase 2 | ||||||
| QLS-111-FDCPOAG/OHT | Phase 2 | ||||||
QLS-111
QLS-111, a novel formulation utilizing our ATP-sensitive potassium (KATP) channel modulator platform, lowers intraocular pressure (IOP) by relaxing vessels of the vascular and vascular-like tissues distal to the Trabecular Meshwork, thereby reducing distal outflow resistance and lowering Episcleral Venous Pressure (EVP).
Though multiple mechanisms of action exist to lower IOP in patients with glaucoma, these agents target only 3 of the 4 components of IOP as described by the Goldmann equation for IOP: the Aqueous Humor Inflow Rate (Q), Uveoscleral Outflow Rate (U), or Conventional Outflow Facility (C). There are currently no approved drugs that selectively target the reduction of EVP. This leaves a significant gap in the potential to maximally lower IOP, since EVP can be the largest determinant of overall IOP.
Pre-clinical and clinical studies have demonstrated that treatment with QLS-111, a unique KATP channel opener, provides persistent IOP lowering without tachyphylaxis, maintains normal vascular integrity of the venous system, and does not cause hyperemia.
QLS-111-FDC
QLS-111-FDC is a novel fixed-dose combination formulation that combines QLS-111 with latanoprost, a gold standard prostaglandin therapy for glaucoma.